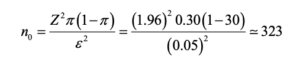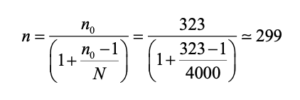Pfupajena Barbara1, Ndaimani Augustine2, Doreen Mukona3, Maxwell Mhlanga4
- Department of Primary Health Sciences, Faculty of Medicine and Health Sciences, University of Zimbabwe, Harare, Zimbabwe.
- Center for Nursing and Midwifery, University of Global Health Equity, Kigali, Rwanda.
- Fatima College of Health Sciences, Department of Nursing, Ajman, United Arab Emirates
- Department of Humanities and Social Medicine, University of Global Health Equity, Kigali, Rwanda
* Corresponding author: Maxwell Mhlanga., Department of Humanities and Social Sciences, University of Global Health Equity. Rwanda. E-mail: mmhlanga@ughe.org
Cite this article
ABSTRACT
Background: SARS-CoV-2 poses a persistent occupational risk to healthcare workers (HCWs) and, by extension, to health professions students undertaking clinical placements. Undergraduate clinical students represent a bidirectional transmission risk potentially carrying infection between campus and clinical environments, yet their specific infection prevention and control (IPC) practices remain poorly characterised in sub-Saharan African academic settings. This study examined IPC practices among health professions students at the University of Zimbabwe during the active phase of the COVID-19 pandemic.
Methods: An analytical cross-sectional survey was conducted between April and July 2021 at the University of Zimbabwe’s Faculty of Medicine and Health Sciences. Using convenience lottery sampling, 320 undergraduate health professions students were enrolled. Data were collected via a researcher-administered structured questionnaire the Clinical Student IPC Practices Questionnaire (CSIPQ) comprising 38 items across five domains: (1) hand hygiene, (2) medical mask use, (3) personal protective equipment (PPE) use, (4) scrubs and clinical wear management, and (5) community IPC conduct. The manuscript was prepared in accordance with the STROBE Statement checklist for cross-sectional observational studies.
Results: The cohort comprised 320 frontline clinical students, predominantly young adults aged 20-25 years (95.3%) with a male preponderance (65.0%). Participants were drawn from five clinical programmes, with Medicine and Surgery (35.6%) and Nursing Science (28.8%) representing the largest groups, and half were in their fourth year of study, indicating advanced clinical exposure.
The prevalence of optimal IPC practice was critically low across all clinical domains: medical mask use (0.6%), PPE use (0.3%), and handwashing (15.3%). Significant associations were observed between handwashing and degree programme (p=0.002) and age (p<.0001). No significant demographic or academic associations were identified for mask use or PPE use, indicating that deficits were uniformly distributed across the cohort.
Conclusion: While specific student demographics were associated with better hand hygiene, near-universal gaps in PPE and medical mask use indicate a systemic failure transcending individual characteristics. Transformative, competency-based IPC education, resource security, and institutional safety culture reform are urgently needed to protect future health professionals and their patients.
Keywords: Health Professions Students; Infection Control; Hand Hygiene; Personal Protective Equipment; COVID-19; Cross-Sectional Studies; Zimbabwe.
INTRODUCTION
The Coronavirus Disease 2019 (COVID-19) pandemic, declared by the World Health Organization (WHO) in March 2020, has posed an unprecedented threat to global health systems, with health care workers (HCWs) bearing a disproportionate burden of infection and mortality [1]. HCWs face a significantly elevated risk, with studies indicating they are over three times more likely to contract SARS-CoV-2 than the general public [2]. This vulnerability extends to trainee HCWs, clinical students who operate at the critical nexus of academic and healthcare settings. As essential participants in patient care, these students are exposed to both community and occupational transmission risks, yet their specific infection prevention and control (IPC) practices remain an underexplored determinant of personal and patient safety [3].
Infection prevention and control (IPC) is the cornerstone of pandemic response, defined by WHO as a practical, evidence-based approach to preventing avoidable infections in patients and health workers [1]. Core non-pharmaceutical interventions, including hand hygiene, respiratory etiquette, and the correct use of personal protective equipment (PPE), are universally acknowledged as critical for breaking chains of transmission [4]. However, adherence to these protocols is influenced by a complex interplay of knowledge, resource availability, institutional policy, and sociodemographic factors [5]. In low-resource settings, which characterize much of sub-Saharan Africa, challenges such as PPE shortages, high-density living conditions, and financial constraints on students can severely compromise ideal IPC practice [6].
Although the WHO declared the end of the COVID-19 public health emergency of international concern on 5 May 2023, SARS-CoV-2 continues to circulate globally and poses an ongoing risk, particularly to elderly individuals and those with comorbidities or disabilities [7]. Furthermore, the clinical sequelae of infection extend beyond the acute phase: Long COVID characterised by persistent fatigue, cognitive impairment, dyspnoea, and multi-system dysfunction lasting weeks to months after infection represents a significant individual and societal burden that reinforces the continued importance of robust IPC practices among all healthcare workers [6].
Vaccination constitutes a primary prevention strategy against SARS-CoV-2 transmission and severe disease. However, studies among nurses and nursing students have identified significant vaccination hesitancy, influenced by social media exposure, misinformation, and attitudinal factors underscoring that immunisation alone is insufficient and must be complemented by strong IPC practice competencies [8 -10].
The context of higher education introduces unique vulnerabilities. Universities are high-density, mobile environments where large gatherings and shared facilities can accelerate outbreaks [11]. When clinical students rotate through healthcare facilities, they become potential bidirectional vectors of SARS-CoV-2, capable of carrying infection from campus to clinic and vice versa [12]. This risk is not merely theoretical; reports from Zimbabwe have documented significant COVID-19 outbreaks among student nurse cohorts within major teaching hospitals [9]. Despite regional and national commitments to strengthen IPC training and PPE provision, compliance gaps persist, suggesting that global guidelines may not adequately address the contextual realities of resource-limited academic and clinical environments [13].
Importantly, the psychological burden of the pandemic on health professions students must be acknowledged. COVID-19 phobia, academic exhaustion, and social isolation have been associated with increased dropout intention among nursing and health sciences students [14]. Fear of infection can erode students’ confidence in clinical environments, contributing to burnout and attrition from health workforce training pipelines further amplifying the importance of adequately protecting students through robust IPC systems.
This study therefore aims to examine SARS-CoV-2 IPC practices among clinical students at the University of Zimbabwe, Faculty of Medicine and Health Sciences. By identifying strengths, gaps, and associated factors, this research seeks to inform targeted, context-specific interventions to better protect future healthcare professionals and the communities they serve.
Objective
The purpose of this study is to assess SARS-CoV-2 Infection Prevention and Control (IPC) practices among undergraduate health professions students specifically those enrolled in Medicine and Surgery, Nursing Science, Physiotherapy, Occupational Therapy, and Radiography at the University of Zimbabwe who had commenced clinical placements.
MATERIALS AND METHODS
Study Setting and Design
This was an analytical cross-sectional survey, prepared in accordance with the STROBE Statement checklist for cross-sectional observational studies [15]. The study was conducted at the University of Zimbabwe, Faculty of Medicine and Health Sciences the oldest and most prestigious university in Zimbabwe, with an enrolment of over 17,000 undergraduate students. The Faculty comprises approximately 23 teaching departments, one School of Pharmacy, and an Institute of Continuing Health Education, offering programmes including Medicine, Dentistry, Pharmacy, Nursing Science, Medical Laboratory Sciences, Rehabilitation, Radiology, and Health Education and Health Promotion. Data were collected between April and July 2021, over 30 weekday data collection sessions. The survey was administered as a researcher-administered, in-person, paper-based questionnaire.
Study Population
The target population comprised undergraduate health professions students who had commenced clinical placements. Eligible year groups were: Years 3–5 for Medicine and Surgery (clinical rotations beginning in Year 3), and Years 2–4 for Nursing Science, Physiotherapy, Occupational Therapy, and Radiography (clinical placements beginning in Year 2). It is important to note that not all eligible students were included participation was opportunity-based, as described below.
Sample Size Determination
The sample size was calculated using Cochran’s formula with finite population correction. The target population consisted of approximately 4,000 undergraduate clinical students. A 95% confidence level (Z = 1.96) and a 5% margin of error (e = 0.05) were selected.
Because no institutional data were available on infection prevention and control (IPC) practices among clinical students, we used an expected prevalence of 30% for “good IPC practices.” This value was informed by previous studies among health professions students in sub‑Saharan Africa, which consistently report low-to-moderate adherence to IPC measures. Banda et al. (2023) found that fewer than one‑third of pharmacy students in Zambia demonstrated good IPC practices [16]; Olum et al. (2020) reported similarly modest practice levels among Ugandan medical students [17]; and Sethi et al. (2021) observed that good IPC practices among Nigerian health professions students generally ranged between 30% and 40% [18]. These findings support the use of p = 0.30 for planning purposes.
Using Cochran’s formula for an infinite population:
Applying the finite population correction for N = 4000:
Thus, the minimum required sample size was 299 participants.
To ensure that this minimum would be met after accounting for non‑response and incomplete questionnaires, we applied a planned inflation about 7%. Therefore, the target sample size was set at 320 students.
Sampling
Convenience lottery sampling was used to select 320 health professions students. During lunchtime sessions in a student common area frequented by students from all eligible programmes, eligible students who were present were invited to draw a card labelled ‘Yes’ or ‘No’ from a container (with replacement). Students who drew a ‘Yes’ card were enrolled until the required sample size was reached. To minimise duplication, all approached students were asked whether they had previously completed the questionnaire; those confirming prior participation were not re-enrolled. No formal participant tracking log was maintained, which is acknowledged as a limitation. Because participation was opportunity-based at lunchtime, students on afternoon clinical rotations or off-campus placements on sampled days were less likely to be represented, constituting a potential source of selection bias.
Eligibility Criteria
The study included all students currently enrolled at the University of Zimbabwe, Faculty of Medicine and Health Sciences, in the fields of Medicine, Nursing Science, Physiotherapy, Occupational Therapy, or Radiography, and who had attended at least one clinical placement since the onset of the COVID-19 pandemic. Students who had experienced prior COVID-19 illness were excluded, as their practices may have been influenced by prior infection experience.
Data Collection
Data were collected over 30 weekday lunchtime sessions between April and July 2021, in the student common area at the University of Zimbabwe, Faculty of Medicine and Health Sciences. Two trained research assistants, supervised by the principal investigator, administered the structured paper questionnaire in person and were available to address any technical difficulties or survey-related questions. No identifying information appeared on completed questionnaires, which were stored in a lockable cabinet accessible only to the research team.
Measurement/Instrument
A single structured questionnaire the Clinical Student IPC Practices Questionnaire (CSIPQ) was used for data collection. The instrument was developed deductively, drawing on the WHO IPC Framework (2020) and the CDC COVID-19 Infection Control Guidance (2021) as theoretical frameworks, and incorporating items adapted from three peer-reviewed questionnaire-based studies on IPC practices among healthcare workers and students [19]; Hossain et al. 2021 [20]; Olum et al. 2020 [17]). A supplementary table listing the reviewed source studies and the elements adapted from each is provided.
The CSIPQ comprised 38 items across two sections: the first section captured four demographic variables (age, sex, degree programme, academic year); the second section contained 38 behavioural items across five IPC domains: hand hygiene (5 items), medical mask use (6 items), PPE use (8 items), scrubs and clinical wear management (7 items), and community IPC conduct (8 items). Items used binary Yes/No responses for frequency-of-behaviour questions and 5-point Likert-type frequency scales (Never to Always) for behavioural habit items. ‘Optimal practice’ within each domain was defined a priori as consistently endorsing all behaviours within that domain consistent with WHO IPC recommendations.
Content validity was assessed by a panel of three subject-matter experts (two infection control practitioners and one nursing education specialist) prior to the pilot study. A Content Validity Index (CVI) of 0.88 was achieved, and two items were revised based on expert feedback. The CSIPQ demonstrated strong content validity, with Item-Level Content Validity Indices (I-CVIs) ranging from 0.83 to 1.00 and a Scale-Level Content Validity Index (S-CVI/Ave) of 0.94, confirming excellent relevance and clarity of the items as assessed by the expert panel. To ensure clarity, relevance, and face validity, the instrument was pre-tested with 10 students at the University of Zimbabwe who met the inclusion criteria (mixed programme composition, minimum Year 2 with clinical exposure); their data were excluded from the main analysis.
The internal consistency of the tool was evaluated using Cronbach’s alpha (α = 0.82), indicating acceptable reliability.
Data Analysis
Data were analysed using the Statistical Package for Social Sciences (SPSS) version 22. Descriptive statistics including absolute frequencies, relative frequencies (percentages), and 95% confidence intervals for proportions (Wilson score method) were used to describe demographic characteristics and adherence to IPC practices across all five domains. The chi-square test or Fisher’s Exact Test (where cell counts were <5) was used to examine associations between demographic and academic variables and IPC practice categories.
For the handwashing outcome where prevalence was 15.3% (n ≈ 41 events) Poisson regression with robust variance estimation was performed to estimate crude Prevalence Ratios (PR) and Adjusted Prevalence Ratios (aPR), with 95% confidence intervals (CI) and p-values. This method is appropriate for common outcomes and avoids the overestimation of relative risk inherent in logistic regression.
For the medical mask use outcome (prevalence 0.6%; n = 2 events) and the PPE use outcome (prevalence 0.3%; n = 1 event), the extremely low event counts precluded reliable regression modelling due to risks of quasi-complete separation and model non-convergence. For these outcomes, Fisher’s Exact Test was used for subgroup comparisons, as recommended for sparse data. In addition, directly calculated unadjusted Prevalence Ratios (simple relative risks computed directly from 2×2 tables, not model outputs) are reported as descriptive effect measures to communicate the uniformity of deficits across subgroups. These directly calculated PRs are mathematically valid regardless of outcome prevalence and are presented for descriptive completeness only, with their limitations explicitly acknowledged in table footnotes. All tests were two-sided; p < 0.05 was considered statistically significant.
Ethical Considerations
The study received ethical approval on 14 June 2021 from the Joint Research Ethics Committee of Parirenyatwa Group of Hospitals and the University of Zimbabwe Faculty of Medicine and Health Sciences (JREC Ref 243/2021).
Written informed consent was obtained from all participants before enrolment. Confidentiality was maintained by anonymising participant data, and access was restricted to authorised researchers.
RESULTS
Socio-demographic characteristics of participants
The demographic characteristics of the 320 frontline clinical students are summarised in Table 1. The cohort was predominantly young, with 95.3% (n=305) aged 20–25 years, and predominantly male (65.0%, n=208). Students were drawn from five clinical programmes, with Medicine and Surgery (35.6%, n=114) and Nursing Science (28.8%, n=92) comprising the largest groups. Half of the participants (50.0%, n=160) were in their fourth academic year, indicating advanced clinical exposure.
Variable Frequency Percentage Age range 20-25 305 95.3 26-30 14 4.4 31-35 1 0.3 Sex Male 208 65.0 Female 112 35.0 Degree Programme Medicine and Surgery 114 35.63 Nursing Science 92 28.75 Occupational Therapy 10 3.13 Physiotherapy 54 16.88 Radiotherapy 50 15.63 Academic year Second 64 20.0 Third 64 20.0 Fourth 160 50.0 Fifth 32 10.0 Total 320 100.0 Table 1. Demographic characteristics of frontline college students (n = 320)
Adherence to specific Infection Prevention and Control (IPC) measures was variable (Table 2). While foundational practices after exposure risks were near-universal, such as hand hygiene after contact with body fluids (99.4%) and consistent mask-wearing in clinical areas (98.1%), critical procedural precautions demonstrated significant gaps.
Less than half of students reported optimal handwashing between patients (53.1%) or before aseptic procedures (88.1%), and only 26.9% consistently used N95 respirators during aerosol-generating procedures.
In community settings, personal hygiene was prioritized (77.5%), but avoidance of high-risk activities like public transport (9.1%) and social gatherings (27.2%) was low.
Frequency Percentage Handwashing Before and after touching a patient 205 64.1 Wash hands with soap for at least 20 seconds 114 35.6 Before aseptic procedures 282 88.1 After contact with body fluids 318 99.4 Between Patients 170 53.1 Consistent use of a medical mask Always wear a mask when in the clinical area 314 98.1 Always remove when feeding but wear a new one before resuming my shift 40 12.5 Never pull my mask down to rest around my chin 80 25.0 I never wear a single mask for more than one day 62 19.3 I have never washed or reused a surgical mask 194 60.6 Always wear an N95 respirator when participating in aerosol-producing procedures 86 26.9 Use of §PPE Always wash/disinfect my hands before gloving 105 32.8 Always change gloves between patients 264 82.5 Use two pairs of gloves for routine procedures 91 28.4 Always wear goggles whenever there is risk of splashes from bodily fluids 99 30.9 Always wear an apron/gown if there is risk of splashes from bodily fluids 168 52.5 Always remove gown and apron whenever I leave ward 240 75.0 Always put on fresh §PPE gown, apron whenever I return to the clinical area from breaks or errands 102 31.9 Always use separate shoes for inside and outside the clinical area 47 14.7 Use of scrubs Always wash my scrubs/white coat after each use 73 22.8 Always wash my scrubs/white coat immediately after use 26 8.1 Always store my scrubs/white coat separately in a tightly sealed plastic bag until I can wash them 39 12.2 I always wash my scrubs/white coat separate from other clothes 106 33.1 Always wash my scrubs/white coat in hot water 11 3.4 I use disinfectant when washing my scrubs/white coat 59 18.4 Never visit common areas around campus (the library, canteen, class) in my scrubs/white coat 89 27.8 IPC practices in the community Always pay closer attention to personal hygiene 248 77.5 Always avoid public transport 29 9.1 Always use soap and water or use an alcohol-based disinfectant for hand washing 243 75.9 Always wash/disinfect my hands after leaving public space 160 50.0 I stay at home as much as possible 103 32.2 I avoid shaking hands when greeting others 138 43.1 I avoid hugging when greeting others 77 24.1 I avoid social gatherings 87 27.2 § PPE – personal protective equipment
Table 2. Infection Prevention and Control (IPC) practices of frontline health students (N=312).
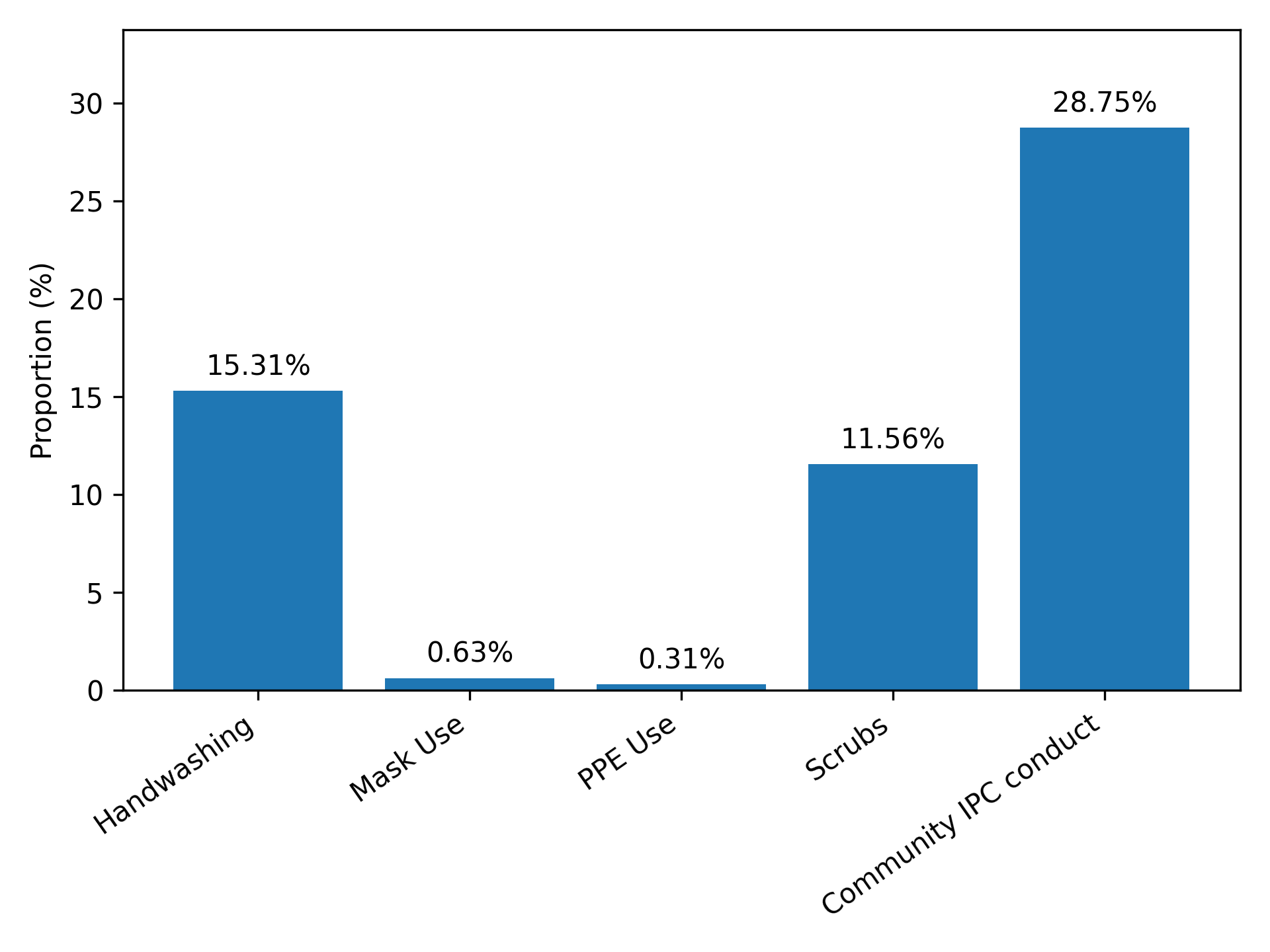
The prevalence of comprehensive adherence to core IPC protocols was critically low across all domains (Table 3). The proportion of students demonstrating optimal practice was highest for community conduct (28.8%, 95% CI: 24.0–34.0), yet fell markedly for essential clinical practices: handwashing (15.3%, 95% CI: 11.8–19.7) and scrubs management (11.6%, 95% CI: 8.5–15.6). The prevalence of correct PPE and medical mask use was exceptionally low, at 0.3% (95% CI: 0.04–2.2) and 0.6% (95% CI: 0.2–2.5) respectively, with confidence intervals indicating these deficits are not due to chance.
Variable Proportion (%)
95% Confidence Interval Lower limit Upper limit Handwashing 15.31 11.75 19.71 Mask Use 0.63 0.16 2.48 PPE Use 0.31 0.043 2.21 Scrubs 11.56 8.48 15.58 Community IPC conduct 28.75 24.03 33.98 Table 3. Prevalence of PPE Use
A composite analysis of optimal Infection Prevention and Control (IPC) practice revealed critically low adherence across all clinical domains (Figure 1).
Figure 1. Number of students who performed optimally on different aspects of IPC
The prevalence of optimal practice was highest for community conduct (28.8%), yet remained below one-third of the cohort. Adherence was markedly lower for essential clinical safeguards: only 15.3% of students demonstrated optimal handwashing, and 11.6% adhered to proper scrubs management. Most alarmingly, the proportion of students performing optimally in correct PPE use (0.3%) and medical mask use (0.6%) was negligible, indicating a near-universal failure to implement these fundamental protective measures.
Association and Regression Analyses by IPC Domain
Mask Use
The analysis of factors associated with optimal mask use revealed no evidence of association between mask use and any demographic or academic subgroup (Table 4).
Variable Frequency *(%) PR (95%CI) aPR (95%CI) p-value Age range 20-25 303 (95.28) Ref. Ref. 26-30 14 (4.40) 1.00 (0.69; 1.46) 1.00 (0.996; 1.01) 0.32 31-35 1 (0.31) 1.00 (0.25; 4.01) 1.01 (0.99; 1.03) 0.31 Sex Female 206 (64.78) Ref. Ref. Male 112 (35.22) 0.996 (0.85; 1.17) 0.99 (0.98; 1.01) 0.31 Degree Programme Medicine and Surgery 113 (35.53) Ref. Ref. Nursing Science 92 (28.93) 0.99 (0.82; 1.21) 0.996 (0.99; 1.00) 0.32 Occupational Therapy 10 (3.15) 1.00 (0.37; 2.69) 1.00 (0.995; 1.00) 0.99 Physiotherapy 54 (16.98) 1.00 (0.80; 1.26) 1.00 (0.998; 1.00) 0.37 Radiotherapy 49 (15.41) 1.00 (0.79; 1.27) 1.00 (0.998; 1.00) 0.55 Academic year Second 64 (20.13) Ref. Ref. Third (19.81) 1.01 (0.79; 1.29) 1.01 (0.99; 1.02) 0.31 Fourth 159 (50.00) 1.01 (0.82; 1.24) 1.01 (0.99; 1.02) 0.31 Fifth 32 (10.06) 1.01 (0.75; 1.36) 1.01 (0.99; 1.02) 0.31 * Row percentage 50% with suboptimal mask use were in fourth year
Table 4. Characteristics associated with mask use among frontline students.
Fisher’s Exact Test showed no significant difference in the distribution of the single optimal-mask-use event across sex, degree programme, or academic year categories (all p > 0.30). Directly calculated unadjusted PRs for all categories approximated 1.00, with 95% confidence intervals crossing the null value, confirming that the critically low prevalence of optimal mask use (0.6%) was uniformly distributed across the cohort. No multivariable adjustment was performed given the insufficient event count (n = 2 events).
Handwashing
The analysis of factors associated with optimal handwashing practice revealed significant associations with specific degree programmes. After adjustment, Occupational Therapy students had a significantly higher prevalence of optimal handwashing compared to Medical students (aPR = 1.08, 95% CI: 1.03–1.14; p = 0.002). A borderline significant association was also observed for Radiotherapy students (aPR = 1.06, 95% CI: 1.001–1.12; p = 0.046). No significant associations were observed for sex or academic year. For the age group 31–35 years, the aPR was 0.55 (95% CI: 0.52–0.58; p < 0.0001), though this estimate is based on a single participant and should be interpreted with extreme caution.
Variable Frequency (%) PR (95%CI) aPR (95%CI) p-value Age range 20-25 259 (95.57) Ref. Ref. 26-30 12 (4.43) 1.00 (0.67; 1.49) 1.06 (0.93; 1.20) 0.40 §31-35 0 (0.00) 0.54 (0.08; 3.85) 0.55 (0.52, 0.58) 0.00 Sex Female 176 (64.94) Ref. Ref. Male 95 (35.06) 1.00 (0.85; 1.19) 0.98 (0.93; 1.03) 0.48 Degree Programme Medicine and Surgery 97 (35.79) Ref. Ref. Nursing Science 72 (26.57) 0.96 (0.79; 1.18) 0.96 (0.89; 1.03) 0.220 § Occupational Therapy 7 (2.59) 1.08 (0.40; 2.91) 1.08 (1.03; 1.14) 0.002 Physiotherapy 47 (17.34) 1.01 (0.83; 1.35) 0.99 (0.93; 1.06) 0.85 §Radiotherapy 48 (17.71) 1.06 (0.83; 1.35) 1.06 (1.001; 1.12) 0.046 Academic year Second 52 (19.19) Ref. Ref. Third 54 (19.93) 1.02 (0.79; 1.31) 0.97 (0.89; 1.06) 0.49 Fourth 140 (51.66) 1.03 (0.83; 1.28) 1.00 (0.94; 1.07) 0.93 Fifth 25 (9.23) 0.98 (0.72; 1.35) 0.94 () 0.31 * Used row percentages; § Statistically significant association
Table 5. Characteristics associated with suboptimal handwashing
PPE Use
The analysis of factors associated with optimal PPE use showed no significant associations with any demographic or academic variable. Fisher’s Exact Test showed no significant difference across any subgroup (all p > 0.30). Directly calculated unadjusted PRs approximated 1.00 with confidence intervals crossing the null, consistent with the interpretation that the critically low prevalence of optimal PPE use (0.31%) was uniformly distributed across the cohort irrespective of student characteristics. No multivariable adjustment was performed given the single-event outcome (n = 1 event).
Variable
Frequency (%)
PR (95%CI)
aPR (95%CI)
p-value
Age range
20-25
304 (95.30)
Ref.
Ref.
26-30
14 (4.39)
1.00 (0.69; 1.46)
1.00 (0.996; 1.01)
0.32
31-35
1 (0.31)
1.00 (0.25; 4.01)
1.00 (0.99; 1.03)
0.31
Sex
Female
208 (65.20)
Ref.
Ref.
Male
111 (34.80)
0.996 (0.85; 1.17)
0.99 (0.98; 1.01)
0.31
Degree Programme
Medicine and Surgery
114 (35.74)
Ref.
Ref.
Nursing Science
91 (28.53)
0.99 (0.82; 1.21)
0.996 (0.99; 1.00)
0.32
Occupational Therapy
10 (3.14)
1.00 (0.37; 2.69)
1.00 (0.995; 1.00)
0.99
Physiotherapy
54 (16.93)
1.00 (0.80; 1.26)
1.00 (0.998; 1.00)
0.37
Radiotherapy
50 (15.67)
1.00 (0.79; 1.27)
1.00 (0.998; 1.00)
0.55
Academic year
Second
63 (19.75)
Ref.
Ref.
Third
64 (20.06)
1.01 (0.79; 1.29)
1.01 (0.99; 1.02)
0.31
Fourth
160 (50.16)
1.01 (0.82; 1.24)
1.01 (0.99; 1.02)
0.31
Fifth
32 (10.03)
1.01 (0.75; 1.36)
1.01 (0.99; 1.02)
0.31
*Used row percentages
Table 6. Characteristics associated with PPE use among frontline students. (AIC=2.68)
In addition, we wish to clarify that the statistical validity of the analysis was not compromised by the missing responses. The minimum required sample size, based on our calculation, was 299 participants. The study targeted 320 students, incorporating an anticipated 7% non‑response rate to ensure that the analytic sample would remain above this threshold. Although eight participants did not complete all questionnaire items, the resulting analytic sample sizes (ranging from 312 to 319 across tables) remained well above the minimum requirement. Therefore, the effective analytic sample remained above the minimum required threshold in all tables, ensuring that no sample‑size–related bias was introduced into the analysis.
DISCUSSION
This cross-sectional study assessed SARS-CoV-2 IPC practices among undergraduate health professions students including Medical, Nursing, Physiotherapy, Occupational Therapy, and Radiography students at the University of Zimbabwe. Few global studies have explored this topic specifically among health professions students in sub-Saharan African contexts, and several have reported suboptimal adherence [11–15]. The present study found a similar pattern, with near-universal deficits in high-stakes clinical IPC practices.
Infection Prevention and Control Practices Among Frontline College Students
Hand Hygiene
Hand hygiene was evaluated using WHO’s five key moments for handwashing. Students reported high overall compliance especially when at risk of contact with blood or body fluids (99.4%). However, compliance was lowest (32.8%) before and after wearing gloves. Only 36% of students washed their hands with soap for at least 20 seconds.
This pattern suggests a positive correlation between perceived infection risk and hand hygiene compliance. Fuller and colleagues, in their study “The Dirty Hand in the Latex Glove,” found hand hygiene compliance decreased by 9% when gloves were worn, supporting our finding [21]. A common misconception exists that gloves are a full substitute for hand hygiene. A similar study found that 38% of medical students were unsure of proper hand hygiene practices when gloves were used [22. In our study, medical and nursing students scored highest on IPC, possibly due to their increased exposure to procedures requiring aseptic techniques.
Being an occupational therapy student was associated with an 8% increase in suboptimal hand hygiene (aPR = 1.08, p < 0.001), likely due to fewer opportunities for performing aseptic procedures. Age also played a role: students aged 31–35 were 45% less likely to demonstrate suboptimal handwashing (aPR = 0.55, p < 0.001), possibly due to greater clinical experience and professional training.
Mask Use
While 98.1% reported wearing masks in clinical areas, only 39.4% used them correctly. A significant proportion (75%) rested masks around their chins while eating or drinking, only 12.5% changed masks between breaks, and 80.7% reused single-use masks. Alarmingly, 39.4% washed and reused disposable masks, similar to findings in Ethiopia where the median duration of single-use mask wear was six days [23]. This contrasts with findings from Poland, where only 24.3% reused single-use masks [24], likely reflecting economic disparities. No demographic variable significantly influenced mask use.
Personal Protective Equipment (PPE)
Consistent PPE use in clinical settings was reported by only 35% of students, higher than the 24.2% reported in a Bangladeshi study [24]. Stockouts and discomfort were identified as barriers, consistent with findings from Tirivavi and others [25]. Proper donning and doffing were practiced by 53.5% of participants, comparable to the Bangladesh study (59.8%), suggesting knowledge gaps in correct PPE use [26]. Reuse of protective gowns was common (68.1%), often due to stockouts.
Fourth-year students had the highest rate of good IPC practices (48.8%; p = 0.02), possibly due to increased clinical exposure. Paradoxically, fifth-year students had the lowest scores (6.9%; p = 0.02). Nursing science students recorded the highest proportion of good IPC practices (34.4%; p = 0.05), likely due to more frequent patient contact and higher perceived infection risk.
This category had the lowest IPC compliance, with only 5.6% reporting consistent practice. Few students washed clinical wear after use (22.8%), washed it separately (33.1%), or used disinfectant soap (18.4%). These figures are lower than those reported in Saudi Arabia, where over half of dental students washed white coats after each use [27]. Limited access to running water in Zimbabwe may explain the low compliance.
Despite low hygiene, 72.2% reported visiting common areas in scrubs/white coats, citing inconvenience in changing attire during short breaks. Demographic variables did not significantly affect white coat/scrub hygiene.
IPC Practices in the Community
Most participants (77.5%) reported improved personal hygiene since the pandemic began, similar to Jordanian medical students (84%) [28]. However, fewer reported increased handwashing (50% vs. 87%), social distancing (24.1% vs. 70%), or avoiding handshakes (43.1% vs. 68.3%). These differences may reflect perceived infection risk and prior experiences with outbreaks. Jordan, having faced SARS (2012) and MERS (2015), recorded higher COVID-19 incidence than Zimbabwe in 2021 (73,305 vs. 4,136 cases/million) [29].
Proximity to the outbreak epicenter also seems to matter. In China’s Henan province, close to Wuhan, 89.7% of healthcare workers adhered to proper IPC [13], while in Ethiopia, compliance was lower at 38.7% [22]. Cultural norms, lockdown enforcement, and public health messaging may explain these differences.
In this study, gender, academic year, and degree program were significant. Female students had better community IPC practices than males (71.4% vs. 28.6%; p = 0.02), consistent with findings from Pakistan [30]. Women are often more hygiene-conscious and socialized to follow rules [31].
Fourth-year students again showed better community IPC practices (41.0%; p = 0.00), a trend supported by studies in Uganda showing older students and health trainees had better IPC compliance [32-33]. Nursing students also had the best community IPC scores (37.3%; p = 0.02), consistent with findings from Ethiopia, where nurses outperformed other healthcare workers in IPC adherence.
Study Implications for Nursing Practice
The findings of this study yield several critical implications for nursing education, clinical training, and institutional policy:
- Targeted IPC Curriculum Integration: The severe deficits in PPE use (0.3%) and medical mask protocols (0.6%) which showed no association with student demographics—point to a universal training failure. Nursing curricula must move beyond theoretical knowledge to include mandatory, simulation-based competency training on the correct donning, doffing, and disposal of PPE. This training should be standardized, recurrent, and include objective assessment before students enter clinical placements.
- Contextualized Education on Resource-Limited Practice: The high rates of PPE reuse and suboptimal scrubs hygiene, heavily influenced by stockouts and lack of facilities, require a pragmatic educational approach. Nursing education should explicitly address IPC adaptations and safety-conscious improvisation for low-resource settings, equipping students to make informed risk assessments without compromising core safety principles.
- Leveraging Professional Socialization and Role Modeling: The finding that Nursing students consistently demonstrated comparatively better IPC practices underscores the potential of professional socialization. Clinical training should intentionally leverage positive peer influence and ensure nursing preceptors exemplify impeccable IPC adherence, as students in high-exposure roles (like nursing) are pivotal in establishing safety norms for interprofessional teams.
- Institutional Accountability for a Safe Learning Environment: The uniform lack of association between student factors and critical IPC failures shifts the onus to institutions. Nursing schools and their affiliated healthcare facilities must co-develop and enforce clear policies guaranteeing consistent access to essential IPC materials (soap, water, PPE) for students. Creating a safety culture where students are not penalized for refusing unsafe assignments due to lack of equipment is paramount.
- Bridging the Community-Clinical IPC Gap: The disparity between higher community hygiene awareness and poor clinical-specific practice indicates a compartmentalization of knowledge. Nursing education should explicitly connect community-based prevention with clinical infection control, framing both as integral components of the professional role. This holistic approach can foster the consistent, context-independent application of IPC principles.
Future research should include: (1) longitudinal studies tracking IPC competency development across clinical years; (2) observational studies using direct observation to validate self-reported practices; (3) intervention trials evaluating simulation-based IPC training programmes in low-resource African settings; and (4) multi-institutional comparative studies across Zimbabwe and sub-Saharan Africa to establish generalisable baseline data for IPC preparedness among future health professionals.
Study Limitations
Several limitations of this study must be acknowledged. First, the cross-sectional design and reliance on self-reported practices limit causal inference and carry risk of social desirability bias; reported adherence likely overestimates actual compliance, which would be better captured by direct observation. Second, the convenience lottery sampling method whereby students self-selected into a common area during lunchtime does not guarantee equal probability of inclusion across all eligible students; those on off-campus rotations or afternoon practicals were likely underrepresented, and no formal mechanism was employed to detect participation bias. Third, data were collected from a single institution, limiting generalisability to other health training institutions in Zimbabwe and the region. Fourth, important potential confounders including prior IPC training quality, individual risk perception, specific clinical rotation exposures, and PPE availability during the survey period were not measured. Fifth, the Clinical Student IPC Practices Questionnaire (CSIPQ) underwent reliability testing (Cronbach’s α = 0.82) but was not subjected to construct validation through exploratory or confirmatory factor analysis; the domain structure is theoretically derived rather than empirically confirmed, and future studies should conduct full psychometric validation of the instrument. Finally, the extremely low prevalence of optimal mask use and PPE use (≤2 events each) precluded multivariable regression modelling for these outcomes; the directly calculated PRs reported for these outcomes are descriptive only and do not represent adjusted estimates.
CONCLUSION
This study reveals a paradox in IPC preparedness among future healthcare professionals at the University of Zimbabwe: while awareness of general hygiene is high, adherence to essential, high-stakes clinical protocols particularly correct PPE and medical mask use is near-universally deficient across all health professions programmes studied. Alarmingly, these critical gaps showed no association with any individual student characteristic, indicating a systemic failure transcending demographics, training year, or programme of study.
The findings underscore an urgent need to move beyond knowledge-based IPC education. Protecting the health workforce pipeline across all health professions including medicine, nursing, physiotherapy, occupational therapy, and radiography demands a transformative approach centred on mandatory simulation-based competency training, guaranteed access to essential resources, and the cultivation of an institutional safety culture where best practice is modelled, enabled, and expected. Given that nursing constitutes the largest component of the frontline clinical workforce, nursing education and professional bodies are particularly well-positioned to champion this systemic change not only in safeguarding nursing students, but in establishing IPC standards that protect patients and the wider health system against current and future infectious threats.
Funding statement
This research did not receive any specific grant from funding agencies in the public, commercial, or not for profit sectors.
Conflict of interest
The authors report no conflict of interest.
Authors’ contribution
Conceptualization: B.P. and A.N; methodology: B.P. and A.N.; software: B.P. and M.M; validation: B.P, D.N, M.M. and A.N.; statistical analysis: M.M., and A.N.; investigation: B.P.; resources: B.P. and A.N.; data curation: B.P. and M.M.; writing- original draft preparation: B.P.; writing-review and editing: A.N.; M.M and D.M; visualisation: M.M.; supervision: A.N.; projection administration: B.P.; funding acquisition: N/A. All authors have read and agreed to the published version of the manuscript.
Acknowledgements
We would want to acknowledge Management for Parirenyatwa Group of Hospitals for allowing us to carry out our research at their institution.
REFERENCES
- World Health Organization (WHO). Infection prevention and control during health care when novel coronavirus (nCoV) infection is suspected: interim guidance, 25 January 2020. Available from: https://www.who.int/publications/i/item/10665-331495
- Alao M, Durodola A, Ibrahim O, Asinobi O. Assessment of health workers’ knowledge, beliefs, attitudes, and use of personal protective equipment for prevention of COVID-19 infection in low-resource settings. Adv Public Health. 2020;2020:1–9. doi:10.1155/2020/6371498
- Assefa D, Melaku T, Bayisa B, Alemu S. Knowledge, attitude and practice of health professional students towards COVID-19 prevention and control in Ethiopia. PLoS One. 2022;17(5):e0268582. doi:10.1371/journal.pone.0268582
- Centers for Disease Control and Prevention (CDC). Infection Control Guidance for Healthcare Professionals about Coronavirus (COVID-19). Atlanta: CDC; 2021. Available from: https://www.cdc.gov/coronavirus/2019-ncov/hcp/infection-control.html
- Moore D, Gamage B, Bryce E, Copes R, Yassi A. Protecting health care workers from SARS and other respiratory pathogens: Organizational and individual factors that affect adherence to infection control guidelines. Am J Infect Control. 2021;49(1):113–119. doi:10.1016/j.ajic.2020.06.213
- Dzinamarira T, Murewanhema G, Musuka G. COVID-19 prevention in Zimbabwe: a review of lessons learnt and way forward. J Community Health. 2021;46(3):668–675. doi:10.1007/s10900-020-00952-3
- Gonzalez B, Mendes TP, Pinto R, et al. Predictors of higher education dropout intention in the post-pandemic era: the mediating role of academic exhaustion. PLoS One. 2025;20(7):e0327643. doi:10.1371/journal.pone.0327643
- Erfani G, McCready J, Nichol B, et al. Identifying patterns and profiles of vaccination hesitancy among nurses for tailoring healthcare policies in the UK: a cross-sectional study. Int Nurs Rev. 2025;72(2):e13035. doi:10.1111/inr.13035
- Keisala J, Jarva E, Comparcini D, et al. Factors influencing nurses and nursing students’ attitudes towards vaccinations: a cross-sectional study. Int J Nurs Stud. 2025;162:104963. doi:10.1016/j.ijnurstu.2024.104963
- Comparcini D, Simonetti V, Totaro M, et al. Clustering vaccine hesitancy and social media use of nurses and nursing students: a cross-sectional study. Appl Nurs Res. 2025;84:151976. doi:10.1016/j.apnr.2025.151976
- Walke HT, Honein MA, Redfield RR. Preventing and responding to COVID-19 on college campuses. JAMA. 2020;324(17):1727–1728. doi:10.1001/jama.2020.20027
- Wilson E, Donovan CV, Campbell M, et al. Multiple COVID-19 clusters on a university campus — North Carolina, August 2020. MMWR Morb Mortal Wkly Rep. 2020;69(39):1416. doi:10.15585/mmwr.mm6939e3
- Mpilo Central Hospital. The epidemiology and lessons of SARS‑CoV‑2, Mpilo Central Hospital, Bulawayo, Zimbabwe. Presented at: RCOG Virtual World Congress 2021; June 2021. https://www.researchgate.net/publication/352258082_The_epidemiology_and_lessons_of_SARS_CoV-2_Mpilo_Central_Hospital_Bulawayo_Zimbabwe
- Castagnetto JM, Hancco-Monrroy DE, Caballero-Apaza LM, et al. Professionalism, emotional wellbeing, and dropout intention in health professions students during the pandemic. Sci Data. 2025;12(1):1259. doi: 10.1038/s41597-025-05508-5.
- von Elm E, Altman DG, Egger M, et al. The Strengthening the Reporting of Observational Studies in Epidemiology (STROBE) statement: guidelines for reporting observational studies. J Clin Epidemiol. 2008;61(4):344-349. doi:10.1016/j.jclinepi.2007.11.008
- Mudenda S, Chizimu J, Chabalenge B, et al. Knowledge, attitude, and practices toward infection prevention and control among undergraduate pharmacy students in Zambia: findings and implications. Antimicrob Steward Healthc Epidemiol. 2023;3(1):e154. doi:10.1017/ash.2023.428
- Olum R, Kajjimu J, Kanyike AM, et al. Perspective of medical students on the COVID-19 pandemic: survey of nine medical schools in Uganda. JMIR Public Health Surveill. 2020;6(2):e19847. doi:10.2196/19847
- Sethi S, Labaran AH, Shenkut M, et al. COVID-19 knowledge and infection prevention practices among health professional students in Nigeria. J Infect Dev Ctries. 2021;15(10):1407-1414. doi:10.3855/jidc.14892
- Iliyasu G, Ogoina D, Otu AA, et al. A multi-site knowledge, attitude and practice survey of Ebola virus disease in Nigeria. PLoS One. 2021;16(1):e0244874. doi:10.1371/journal.pone.0244874
- Biesty L, Meskell P, Glenton C, et al. A QuESt for speed: rapid qualitative evidence syntheses as a response to the COVID‑19 pandemic. Syst Rev. 2020;9(1):1–6. doi:10.1186/s13643‑020‑01475‑3
- Sahiledengle B, Tekalegn Y, Woldeyohannes D. The critical role of infection prevention overlooked in Ethiopia: only one-half of health-care workers had safe practice: a systematic review and meta-analysis. PLoS One. 2021;16(1):e0245469. doi:10.1371/journal.pone.0245469
- Zhang M, Zhou M, Tang F, et al. Knowledge, attitude, and practice regarding COVID-19 among healthcare workers in Henan, China. J Hosp Infect. 2020;105(2):183–187. doi:10.1016/j.jhin.2020.04.012
- Kassie BA, Adane A, Tilahun YT, et al. Knowledge and attitude towards COVID-19 and associated factors among health care providers in Northwest Ethiopia. PLoS One. 2020;15(8):e0238415. doi:10.1371/journal.pone.0238415
- Mani NS, Budak JZ, Lan KF, et al. Prevalence of Coronavirus Disease 2019 infection and outcomes among symptomatic healthcare workers in Seattle, Washington. Clin Infect Dis. 2020;71(10):2702–2707. doi:10.1093/cid/ciaa761
- Fuller C, Savage J, Besser S, et al. ‘The dirty hand in the latex glove’: a study of hand hygiene compliance when gloves are worn. Infect Control Hosp Epidemiol. 2011;32(12):1194–1199. doi:10.1086/662619
- Al Kadi A, Salati SA. Hand hygiene practices among medical students. Interdiscip Perspect Infect Dis. 2012;2012:1–6. doi:10.1155/2012/579631
- Khasawneh AI, Humeidan AA, Alsulaiman JW, et al. Medical students and COVID-19: knowledge, attitudes, and precautionary measures. A descriptive study from Jordan. Front Public Health. 2020;8:253. doi:10.3389/fpubh.2020.00253
- Matusiak L, Szepietowska M, Krajewski PK, et al. The use of face masks during the COVID-19 pandemic in Poland: a survey study of 2315 young adults. Dermatol Ther. 2020;33(6):e13909. doi:10.1111/dth.13909
- Hossain MA, Rashid MUB, Khan MAS, et al. Healthcare workers’ knowledge, attitude, and practice regarding personal protective equipment for the prevention of COVID-19. J Multidiscip Healthc. 2021;14:229. doi:10.2147/JMDH.S293717
- Tirivavi, E., Chikanya, V., & Mundagowa, P. T. (2019). Infection control practices associated with puerperal sepsis in Harare City Maternity Units. Central African Journal of Public Health, 5(1), 31–35. doi:10.11648/j.cajph.20190501.15
- Subramaniam, T., Loo, R. C. N., & Poovaneswaran, S. (2013). The practice of PPE amongst fourth year medical students at A&E. Where are we? International Medical Journal Malaysia (IeJSME), 7(2), 29–32. doi:10.56026/imu.7.2.29
- Aljohani Y, Almutadares M, Alfaifi K, et al. Uniform-related infection control practices of dental students. Infect Drug Resist. 2017;10:135. doi:10.2147/IDR.S126631
- Lai X, Wang X, Yang Q, et al. Will healthcare workers improve infection prevention and control behaviors as COVID-19 risk emerges and increases, in China? Antimicrob Resist Infect Control. 2020;9(1):1–9. doi:10.1186/s13756-020-00760-1
This work is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License.